Our Solutions
Designed for cross-functional teams who see where work gets stuck and want a practical way to fix it.

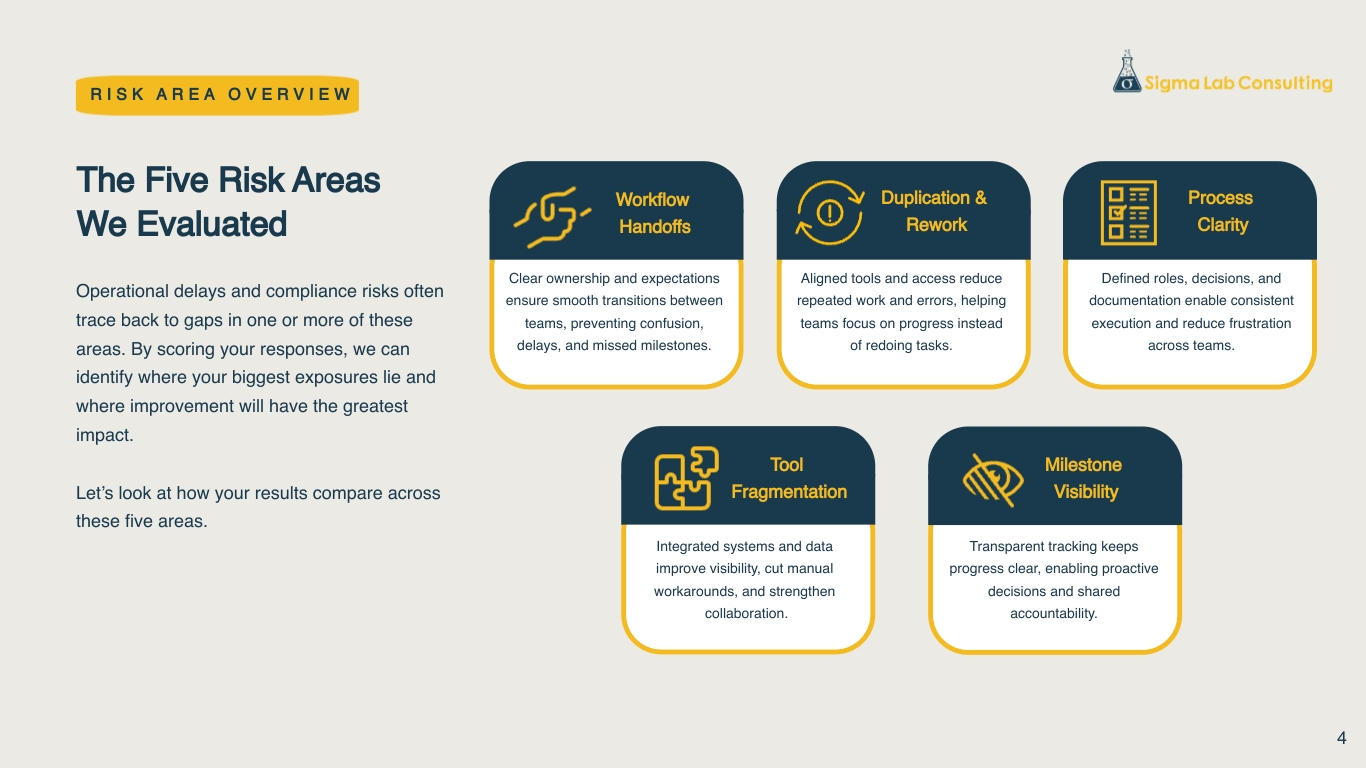
Why This Works
Our Operational Risk Assessments cut through noise and uncertainty by centering on the real decisions your team needs to make — not a generic diagnostic. The hybrid, fully virtual experience gives participants space to reflect honestly on where work gets stuck, while our transparent, proven framework turns that insight into a clear picture of risk and 3–5 practical moves for the next quarter. It feels like a 1:1 working session, without demanding time your team doesn’t have, and delivers value whether or not you engage us further.

Modules & Assessments
1.1 Equipment Onboarding Strategy
Goal:
Ensure new equipment, systems, and facilities are onboarded efficiently and compliantly by identifying and eliminating friction points that cause delays, misalignment, or compliance risk.
Why This Matters:
Every delay or compliance issue in startup can usually be traced to a breakdown in equipment onboarding. By scoring your responses against this framework, we can quickly pinpoint your exposures.
1.2 Facility Maintenance Strategy
Goal:
Evaluate whether your maintenance practices protect uptime, compliance, and operational agility.
Why This Matters:
Unplanned downtime is the symptom — not the problem. This assessment reveals the maturity gaps in your maintenance strategy and how to close them before they cost you.
2.1 Operational Workflow Alignment
Goal:
Discover inefficiencies in how core scientific, research, or technical teams interact with business and support operations.
Why This Matters:
Your teams are busy — but not aligned. This assessment identifies the handoff, clarity, and integration breakdowns holding your system back.
2.2 Scientific Onboarding & Offboarding Risk
Goal:
Reduce knowledge loss and operational risk tied to personnel transitions in technical roles.
Why This Matters:
Every transition risks losing knowledge. This assessment surfaces the onboarding and offboarding gaps that weaken your research continuity.
3.1 Cross-Functional Project Execution Health
Goal:
Diagnose how consistently teams deliver complex, multi-stakeholder projects.
Why This Matters:
Complex projects demand more than hard work — they demand execution discipline. This assessment pinpoints the scoping, governance, and communication gaps putting your delivery at risk.
3.2 Decision-Making & Governance Flow
Goal:
Improve speed, transparency, and accountability in how decisions are made and communicated.
Why This Matters:
Slow decisions are costly decisions. This assessment uncovers the governance and decision flow gaps slowing you down.
4.1 Digital Operations & Data Flow
Goal:
Uncover breakdowns in how data moves between systems, teams, and functions.
Why This Matters:
Your digital systems should speed you up — not slow you down. This assessment pinpoints integration gaps and bottlenecks in your digital operations.
4.2 Role-Based Training & Competency System
Goal:
Ensure the organization can demonstrate readiness, compliance, and adaptability through its people.
Why This Matters:
If your people aren’t ready, your organization isn’t ready. This assessment finds the training and competency gaps that put compliance and agility at risk.
4.3 Regulatory Interface & Submission Risk
Goal:
Improve the operational handshake between those who generate data (clinical, product, R&D) and those who report it (regulatory).
Why This Matters:
Late-stage submission risk is avoidable. This assessment exposes the planning, documentation, and change control gaps that can derail approval timelines.
Each assessment is designed to deliver rapid clarity.
For teams ready to take the next step, we build on these insights through targeted consulting and implementation support, helping you design solutions that stick and drive measurable impact.


FAQs
Most participants spend about 45–60 minutes. You can pause and return anytime within 30 days.
Our team reviews your responses and prepares a customized report highlighting your top operational exposures and recommendations.
Reports are shared during your 1:1 debrief session, usually within 72 hours of submission.
A 45–60 minute session with a Sigma Lab expert to review results, clarify findings, and discuss next steps.
Yes, and we recommend it—especially those in Operations, Quality, or R&D.
Absolutely. All assessment data and discussions are confidential. (See our Privacy Policy for details.)
Yes. Many clients complete multiple assessments across different focus areas.
Contact admin@sigmalabconsulting.com for quick support.
No data gathering needed—just reflect on your current workflows and challenges.
A customized report and actionable insights to reduce risk and improve alignment.
Payments are processed securely through Stripe, a globally trusted, PCI-compliant payment platform. Sigma Lab Consulting does not store or process credit card information directly and maintains strict standards for data security, confidentiality, and financial integrity.
We accept major credit and debit cards via Stripe. For organizations that require payment via purchase order (PO) or prefer to be invoiced, please contact admin@sigmalabconsulting.com, and our team will coordinate the appropriate payment process.




